Abbott's FreeStyle® Libre 2 iCGM Cleared in U.S. for Adults and Children with Diabetes, Achieving Highest Level of Accuracy and Performance Standards
Por un escritor de hombre misterioso

FDA clears Abbott's FreeStyle Libre 2 system in U.S. for adults and children (ages 4 and older) with diabetes, the only iCGM with unsurpassed 14-day accuracy that measures glucose every minute with optional real-time alarms.
/PRNewswire/ -- Abbott (NYSE: ABT), the worldwide leader in continuous glucose monitoring (CGM), announced today the U.S. Food and Drug Administration (FDA)

Abstracts from ATTD 2017 10th International Conference on Advanced Technologies & Treatments for Diabetes Paris, France—February 15–18, 2017
Abbott Releases the FreeStyle Libre 2 iCGM - Children with Diabetes

Surge in FDA Approvals for Diabetes Devices and Product Advances

Cecilia Yats M.A.,R.D.N.,L.D. posted on LinkedIn

PDF) A head-to-head comparison between Guardian Connect and FreeStyle Libre systems and an evaluation of user acceptability of sensors in patients with Type 1 diabetes

New Diabetes Technology Coming in 2022
Freestyle Libre 2 CGM, Strategy and Strength
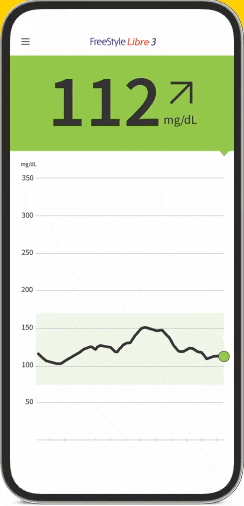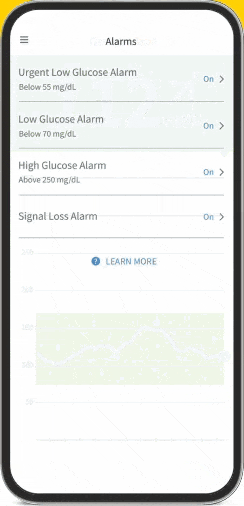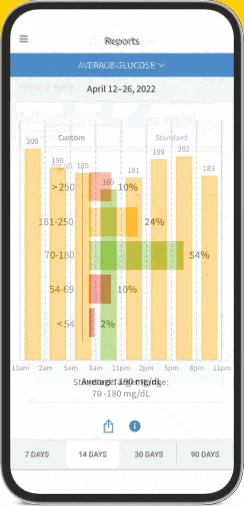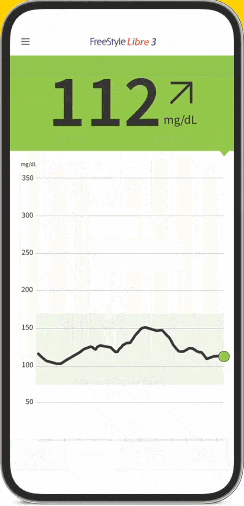
FreeStyle Libre Continuous Glucose Monitoring

Benefits and Limitations of MARD as a Performance Parameter for Continuous Glucose Monitoring in the Interstitial Space. - Abstract - Europe PMC
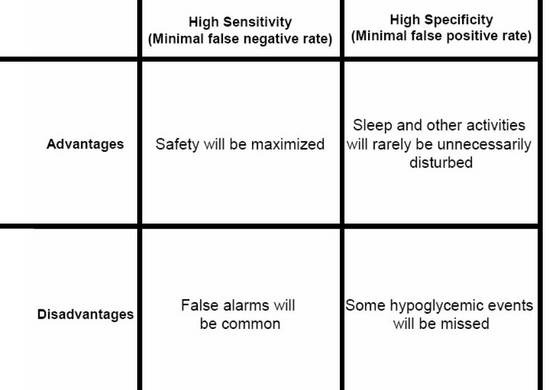
Monitoring Technologies- Continuous Glucose Monitoring, Mobile Technology, Biomarkers of Glycemic Control - Endotext - NCBI Bookshelf

Effect of 6 months' flash glucose monitoring in adolescents and young adults with type 1 diabetes and suboptimal glycaemic control: managing diabetes in a 'flash' randomised controlled trial protocol

FreeStyle Libre 2 Continuous Glucose Monitor
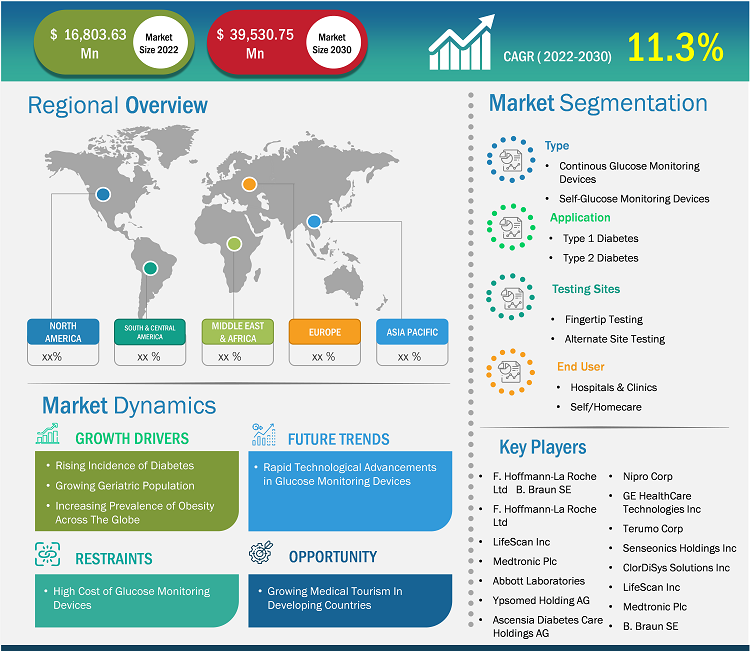
Glucose Monitoring Devices Market Scope Report

Buy Freestyle Libre Two Sensor, Glucose Monitor

Diabetes Connections, Type 1 Diabetes, Podcast