Abbott's FreeStyle® Libre 2 iCGM Cleared in U.S. for Adults and Children with Diabetes, Achieving Highest Level of Accuracy and Performance Standards - Jun 15, 2020
Por un escritor de hombre misterioso
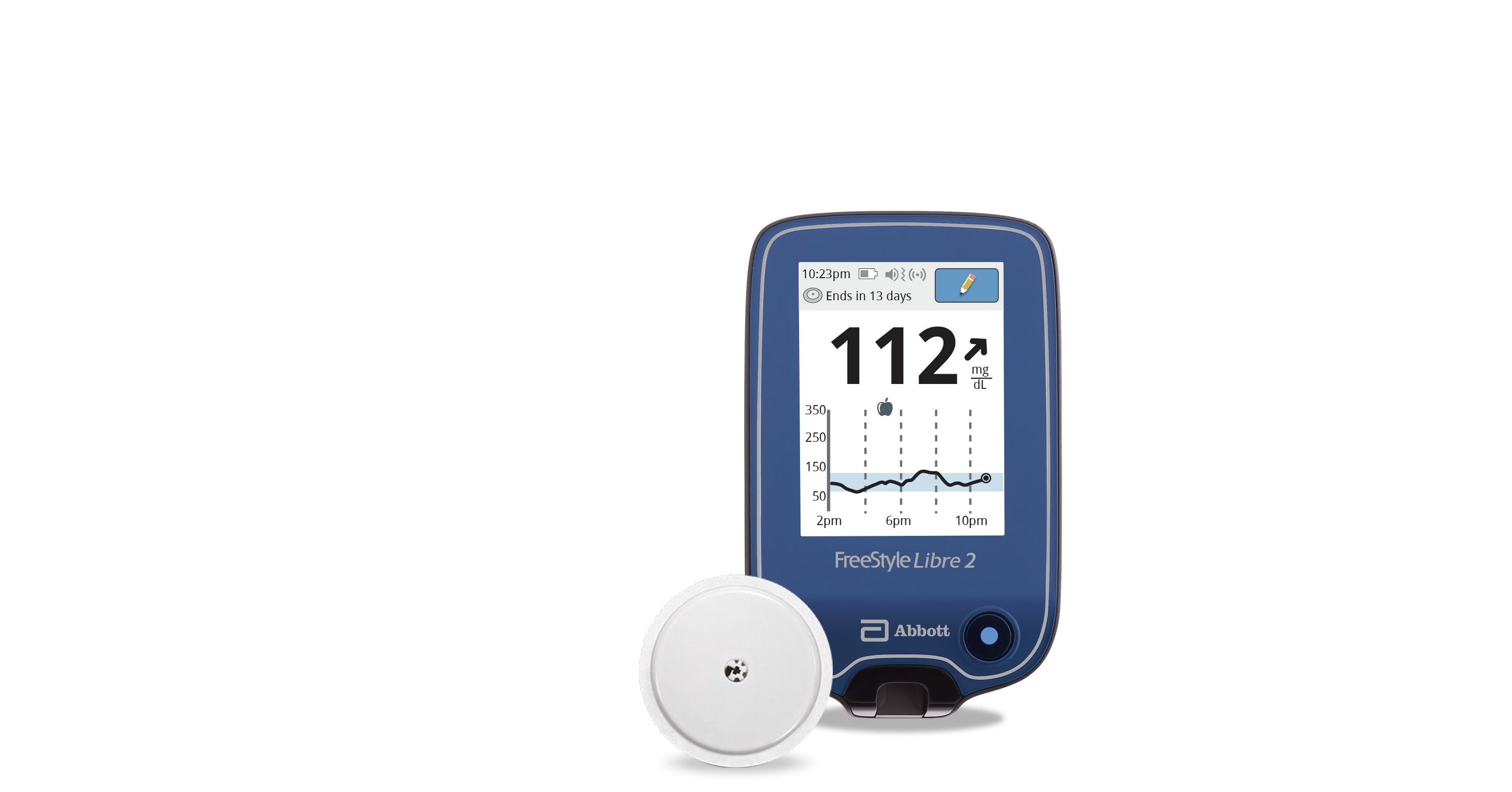
Abbott (NYSE: ABT), the worldwide leader in continuous glucose monitoring (CGM), announced today the U.S. Food and Drug Administration (FDA) cleared its next-generation FreeStyle® Libre 2

Improving Equitable Access to Continuous Glucose Monitors for Alabama's Children with Type 1 Diabetes: A Quality Improvement Project

FDA Approves FreeStyle Libre 2 iCGM System for Patients With Diabetes
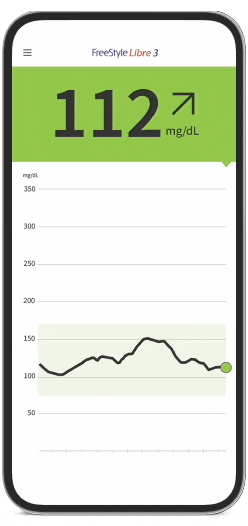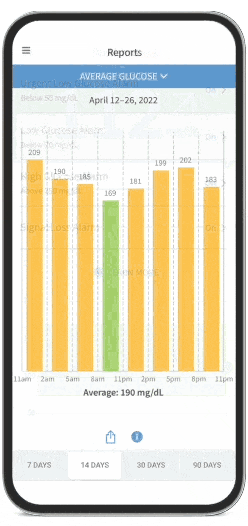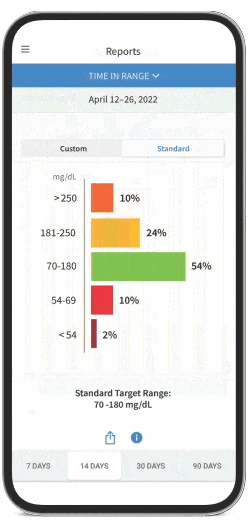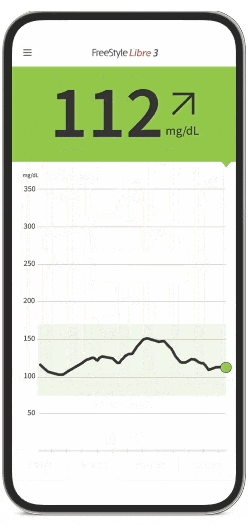
FreeStyle Libre Continuous Glucose Monitoring

FreeStyle Libre 2 System
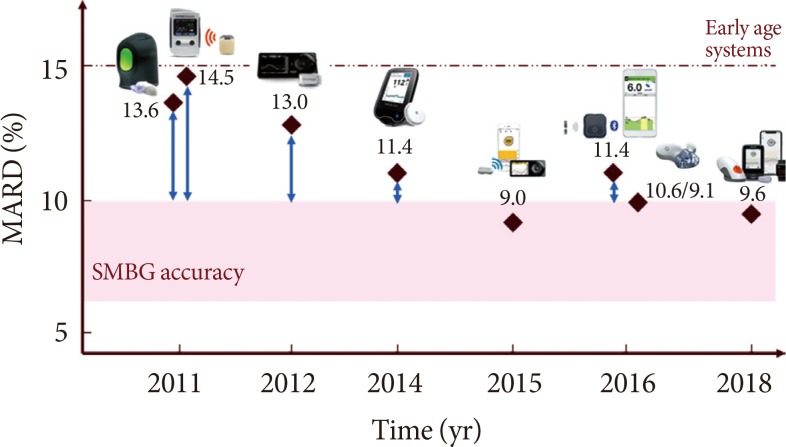
Continuous Glucose Monitoring Sensors for Diabetes Management: A Review of Technologies and Applications

FreeStyle Libre 2 Continuous Glucose Monitor

FreeStyle Libre 2 Continuous Glucose Monitor

2 Million Patients And Beyond: Abbott's FreeStyle Libre 2 Cleared In The U.S. For Adults And Children With Diabetes
New Study Published in The New England Journal of Medicine Demonstrates Abbott's FreeStyle Libre® 2 System Has Positive Impacts on Glucose Levels and Quality of Life - Oct 6, 2022

Landscape of Continuous Glucose Monitoring (CGM) and Integrated CGM: Accuracy Considerations

Abbott's FreeStyle® Libre 2 iCGM Cleared in U.S. for Adults and Children with Diabetes, Achieving Highest Level of Accuracy and Performance Standards

Abbott's FreeStyle Libre 2 CGM picks up Medicare coverage
FreeStyle Libre 3: World's Smallest Sensor